The Healthcare Paradox: An Open Letter to Industry Outsiders
Startups don’t enter healthcare; they collide with it. For most, this isn’t a strategic expansion — it’s an emotional abduction triggered by a personal medical crisis. You see a broken system and decide to disrupt it, but you’re bringing a tech knife to a systemic gunfight.
If you want to see how that collision feels from the outside, watch Kamikaze_Shortbus’s video You Don’t Hate the American Healthcare System Enough. If you want to see the cost of trying to “fake it until you make it” in a world of blood and bone, look at Elizabeth Holmes in the HBO Documentary The Inventor: Out for Blood in Silicon Valley. Her downfall started long before the fraud; it began the moment she decided medical standards were just “obstacles” to be willed away. She tried to force a vision into a clinical reality that does not bend to sheer force of will.
But the real reason we are here is because as an engineering partner, I’m tired of seeing amazing ideas fail because of avoidable “outsider” hubris. Nothing saddens me more than seeing a life-saving product die in the dark because a team underestimated the system’s gravity. The system is slow for a reason, but being informed is the only way to move fast without breaking yourself.
Section 1. The FDA is a Shield (The Blood-Written Rules)
Before diving into the systemic business issues that sink outsiders, we have to establish the baseline: the stakes of the medical field are written in blood. This gets forgotten a surprising amoutn of times.
Founders often categorize the FDA as the bureaucratic final boss standing between them and their Series A. However, the agency is better understood as a shield forged from historical trauma. One of the most sobering stories I carry with me — and one I suggest you watch in Kyle Hill’s deep dive, “History’s Worst Software Error” — is the tragedy of the mid-1980s, where an unconstrained “move fast” mentality created catastrophic vulnerabilities.
The Lesson of the Therac-25: The Therac-25 relied on software for safety rather than mechanical interlocks. A race condition meant that if an operator corrected settings in under 8 seconds, the software failed to update the hardware. The machine would fire a high-power electron beam directly into the patient without the “target” required to convert it to X-rays. The console displayed only a cryptic “MALFUNCTION 54”, leading operators to reset and fire again, delivering multiple lethal doses.
This disaster occurred because engineers applied unvetted technology to patients without a safety-first architecture. When you integrate regulatory standards into your workflow, the FDA becomes a silent design partner. Their requirements are the logic that should inform your design requirements from day one. Every filing and documentation phase is geared toward making your product more resilient. Building with these regulations as your scaffolding is the only way to build for healthcare; anything else is simply building a liability.
Section 2. The Engineering Trap: Building a Clinical Fix for a Financial Ghost
Hiring an engineering firm that treats healthcare like a standard technical puzzle is the fastest way to derail a startup. Outside firms bring a mentality suited for consumer goods — like a smart soap dispenser — where the risks of failure are negligible. In a medical context, the physics change the stakes. A low DC voltage is harmless on the skin, but that same current becomes a fatal conductor when applied directly to the heart during an invasive procedure. Standard firms prioritize rapid iteration to get products into “users’ hands,” ignoring that these users are patients who cannot be subjected to a “trial and error” development phase.
Because of this, I advise my customers to seek out firms with a proven history of working within FDA frameworks and a safety-critical background that covers all aspects of the lifecycle. You need a partner that understands how a design choice in month two impacts a filing in year two. Look for teams willing to move in deliberate, bite-sized pieces. Treating the project like a buffet where you de-risk each individual stage prevents the catastrophic rework that occurs when regulators finally audit a rushed design. This requires defined agility: a disciplined iteration process that embeds regulatory guardrails into every sprint.
Engineers from outside the sector also struggle with the industry’s inherent lack of transparency. In most fields, solving a technical snag involves reviewing a map of the entire system. In healthcare, you operate in a fog. You may understand the immediate problem, but the broader ecosystem is gated behind defenses designed to protect patient safety and institutional brands. Access to deeper system layers is earned through proven compliance, not granted by default.
If your engineering team expects open access to a hospital’s backend or a payer’s logic, they are building for a reality that doesn’t exist. Without a deep grasp of HL7 and the specific, siloed implementations of FHIR, your team is simply building an isolated island. If the technical requirements aren’t informed by these systemic barriers from day one, your engineers are front-loading your failure rather than accelerating your launch.The Environmental and Ethical Oversight
Section 3. The ICP Hydra: You Don’t Know Who Your Customer Is
Accelerators love to talk about the Ideal Customer Profile (ICP), but in healthcare, that profile is a three-headed monster. You can build a legitimate breakthrough — like the infusion wedge designed to stabilize needles during phlebotomy — and still watch it die on the floor. On paper, that product was brilliant; the founder raised capital and sold thousands of units. However, once the product reached the hospital, the functional workflow killed it. The wedges were stored in a cabinet too far from the point of care, and clinicians simply went back to folding over cotton gauze because it was already in their hands.
Beyond the workflow, there is a harder question most startups fail to answer: Who is saving or making money because of this?
In the U.S. medical system, your product’s value is usually viewed through two lenses: cost-avoidance or revenue-generation. You are either removing a cost to protect existing profits (e.g., reducing readmissions or infection rates), or you are adding a cost that allows the facility to bill more for a procedure. If your device is just “better” but doesn’t clearly move the needle on one of those two financial levers, it is a non-starter.
The commercial sector allows for “nice-to-have” products if the user likes them. Healthcare does not. The patient cannot access your device unless a doctor recommends it, and a doctor won’t recommend it if it adds thirty seconds of friction to a task they perform fifty times a day. Even then, the hospital won’t stock it unless the Payer has cleared the expense. If you haven’t identified exactly who is losing money by not having your device, or how your device creates a new billable event, you haven’t found your ICP.
Section 4. Visceral Proof: Beyond the Polished Deck
In the commercial tech world, your seed round is often built on a high-polish slide deck and a “paper test” that proves the concept could work. In the medical realm, the industry demands a much grittier form of validation. Whether it is a clinical trial, an animal study on a pig, or even a bench test using a dead chicken leg, stakeholders want to see the product surviving the chaos of a real medical environment.
At a recent medical startup event, I saw this contrast in real time. While most tech founders hide behind “perfect” UI mockups, a neuro-monitoring startup showed raw footage of an open brain surgery. They demonstrated exactly how their model mapped electrical synapses and physical tumor displacement compared to the current standard of care. It wasn’t “pretty,” but it was visceral proof. Medical stakeholders don’t want to see a 100% finished, injection-molded housing; they want to see that in the high-stakes moment of a surgery or a diagnosis, the core technology functions exactly as promised.
This is exactly why so many successful medical startups have practicing clinicians on their boards. These doctors aren’t just there for the pedigree; they are there to testify to the workflow. When a surgeon joins your board, they are putting their reputation on the line to say: “I have seen this device, and I would actually use it in my OR.” Founders often make the mistake of spending their limited runway trying to make their prototype look like a finished consumer product. In reality, you are better off spending that money on a “rough” but functional pilot or a cadaver study that provides that visceral “aha” moment. If a doctor can’t visualize your product covered in fluids and functioning under the stress of a 12-hour shift, your polished slide deck won’t save you. You don’t need perfection; you need to prove that you can handle the reality of the human body.
Section 5. Visceral Proof: Beyond the Polished Deck
In the commercial tech world, your seed round is often built on a high-polish slide deck and a “paper test” that proves the concept could work. In the medical realm, the industry demands a much grittier form of validation. Whether it is a clinical trial or a bench test using animal tissue, stakeholders want to see the product surviving the chaos of a real medical environment.
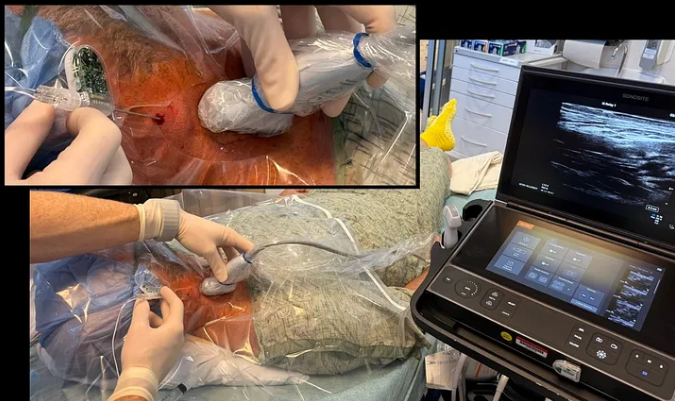
Doctors are trained skeptics; they deal with the physical failures of the human body every day. Showing them visceral images — even unpolished ones — validates that you understand the reality of their work. A prime example of this “clinical-first” proof can be found in a recent case report in the Journal of Orthopaedic Experience & Innovation: “A Novel Percutaneous Device Enabling both Local Anesthetic Delivery and Electrical Stimulation…”.
The “proof” in this study wasn’t a sleek marketing video; it was raw ultrasound guidance images showing the device lying adjacent to the C5 and C6 nerve roots. It documented the specific milliamps used to achieve a “pleasant massage” sensation and detailed the mechanical “tines” used to anchor the catheter. For a startup, this level of detail is much further than a standard prototype, but it is exactly what a clinical buyer expects. They want proof that in the high-stakes moment of a procedure, the device functions within their specific workflow.
This is why successful medical startups prioritize clinicians on their boards. When a surgeon joins you, they are testifying that they have seen the device and would trust it in their own OR. Founders often waste their runway trying to make a prototype look like a finished consumer product, but you are better off spending that money on a functional cadaver study or a technical feasibility report.
If a doctor can’t visualize your product functioning under the stress of a 12-hour shift, your polished slide deck won’t save you. You don’t need perfection; you need to prove you can handle the human element.
Section 6. The Hidden Costs of Purposeful Complexity
Outside founders often fall into the trap of thinking they just need to build a “better” version of what’s already out there. I constantly hear people complaining about Epic — how the UI is clunky and feels like it’s from 1995.
What they don’t realize is that Epic isn’t trying to win a design award; it’s trying to manage the Herculean task of projecting complex, multidimensional data into a 2D world while maintaining a perfect audit trail.
If you think you’re going to win because your app is “sexier,” you’ve already lost. In healthcare, “better” is not an aesthetic choice; it’s a functional definition rooted in practical evidence, not anecdotal ease of use.
The Moat of Reliability: Modern UIs are built for speed and delight, but medical systems are built for consistency and risk mitigation. A “modern” interface that hides a critical alert behind a clean, minimalist menu is a liability. Incumbents use complexity as a defense mechanism because that complexity accounts for the edge cases that actually happen on a hospital floor.
The Integration Tax: Purposeful complexity also serves as a defensive moat. Look at anyone who has tried to integrate with PointClickCare (PCC). They receive thousands of requests a month from developers who “just want access.” PCC’s barrier isn’t a lack of technical support; it’s a demand for proof. They often won’t even talk to you until you can prove you already have a customer. To them, a paying customer is the only valid proof that you are designing something functional rather than just playing with data.
Planning for the Tax: Successful startups don’t get frustrated by this tax; they plan for it. They find clever ways to work within the constraints rather than fighting them. I’ve seen smart EHR plugins that bypass the nightmare of official API negotiations by “scraping” or grabbing data directly from the front-end interface. They understand that getting to market via a functional workaround is better than dying in a three-year queue for an official integration.
The cost of entry isn’t just your R&D; it’s the “tax” of navigating a system that values survival over sexiness. If you don’t account for the time and capital required to hack through this thicket of purposeful complexity, you’ll find yourself with a gorgeous product and a bankrupt company.
Conclusion: Stewardship Over Disruption
The healthcare industry doesn’t need “disruptors” who want to move fast and break things; it needs stewards who respect the gravity of the system. Success in this space requires a rare combination of technical agility and regulatory discipline.
I often look back at a sentiment Mark Cuban is cited for: not every industry needs to be disrupted. This is especially true in healthcare. We don’t need to shatter the foundations; we need to reinforce them. You must be willing to move in bite-sized pieces, de-risking your engineering at every step and treating the FDA as a design partner rather than a hurdle.
If you look at the ideas that actually make it through the gauntlet, they aren’t the most complex — they are the most primitive. Simplicity beats all else. The winners are the ones who solve a basic, necessary need so effectively that the risk of adoption becomes negligible.
You have to look past the “clunky” interfaces of the incumbents to understand the life-saving logic and financial structures that keep them in place. Most importantly, you have to remember that at the end of every data string, every billing code, and every engineering sprint, there is a patient. If you can navigate the purposeful complexity without losing sight of the visceral proof required to gain a clinician’s trust, you won’t just build a product — you’ll build a legacy.
In medical, you don’t win by being the loudest or the prettiest; you win by being the one who is still standing when the fog clears, because you respected the rules written in blood.
About Landi Industries We help teams navigate the complexities of product development by sharing the hard-earned lessons of hardware engineering. From structural design to system architecture, our goal is to provide the roadmap for turning ambitious concepts into reliable physical products.
Stay in the Loop:
The Landi Handbook (Beehiiv): Our primary deep-dive newsletter for tactical engineering advice and hardware development strategies.
LinkedIn Newsletter: A medical-focused newsletter breaking down complex technical topics for non-technical professionals and stakeholders.
YouTube: Visual guides and technical walkthroughs covering the realities of the product development process.
Medium: Long-form articles exploring the deep "why" behind successful hardware and system design.
Work With Us: Need expert help with your next build? See our full range of services at LandiIndustries.com